Evaluating Cell Separation Techniques
Qualities of an Outstanding Cell Separation Method
Cell separation techniques are commonly used when researchers dealing with complex biological samples have a need to isolate and analyze a specific cell type of interest. While deciding which technique used for cell separation best suits a researcher’s needs can be complicated, there are several factors that are routinely used to guide those decisions. Here we summarize those factors and talk about the role that each play when choosing the optimal cell separation method.
What Is Cell Purification?
Since the goal of any cell separation technique is to isolate a particular cell type of interest so that it may be analyzed independently, one of the most important factors to consider when selecting a separation method is purity.
Purity is a measure of how many isolated cells are of the cell type of interest vs. how many belong to undesired cell types. Most often, purity is conveyed as a percentage of desired cells out of the total number of isolated cells. For example, a cell separation having a purity of 90% indicates that 90 out of every 100 cells are the cell type of interest while only 10 out of the 100 are contaminating cell types.
What is considered sufficient cell purity can vary depending on the goals of the experiment, the overall complexity of the sample, and the specificity of the characteristic being used to isolate the target cells. However, greater purity means that the results obtained from a cell separation are more likely attributable to the cell type of interest.
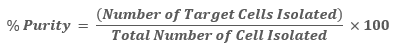
What Is Cell Yield and Recovery?
While purity is important for clarity of results, having a highly pure cell separation is meaningless if you cannot isolate enough cells to accommodate your downstream analysis goals. For these reasons, it is important to also consider yield when choosing a separation technique. Yield is used to describe how many cells of interest were successfully isolated following cell separation. Higher yields provide more cells for your desired applications.
Another way to think about the number of cells isolated during a given cell separation is by calculating recovery. While yield looks at the absolute number of cells isolated, recovery takes into account how many cells of interest were in the starting material and reveals the percentage of those cells that were successfully captured during your separation. This gives researchers an indication of how effective their separation technique is and how many target cells are missed during their selection. Therefore, recovery can be a critical parameter to consider when selecting or optimizing a cell separation method.
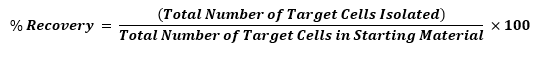
Yield and recovery are intimately connected and can be impacted by numerous factors. For best results, it is critical to start with a fresh sample and use a cell isolation kit of superior quality.
What Is Cell Viability?
Cells that have died or been damaged during the cell separation process spill their contents and expose the neighboring cells to an array of factors that could produce undesirable effects. Those effects could include changes in growth, activation of stress response pathways, initiation of cell death programs, and other alterations in their normal physiology that make obtaining meaningful data from downstream applications challenging. Therefore, the health of isolated cells is essential for ensuring success and is particularly important in cases where live cells are required. For instance, when assessing cellular function or simply culturing the isolated cells.
The most common way of assessing cellular health is by determining viability. Viability is merely a measurement of the proportion of isolated cells that are living after the cell separation procedure. Dyes that enable evaluation of membrane integrity are frequently used for this purpose. Membrane impenetrate dyes such as trypan blue, DRAQ7TM, 7-AAD, PI (Propidium iodide), or DAPI (4′,6-diamino-2-phenylindole, dihydrochloride) can only stain dead or dying cells with damaged membranes. Isolated cells exposed to these dyes can be evaluated with a flow cytometer, microscope, or cell counter to determine overall viability.
Viability is calculated as follows:
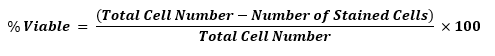
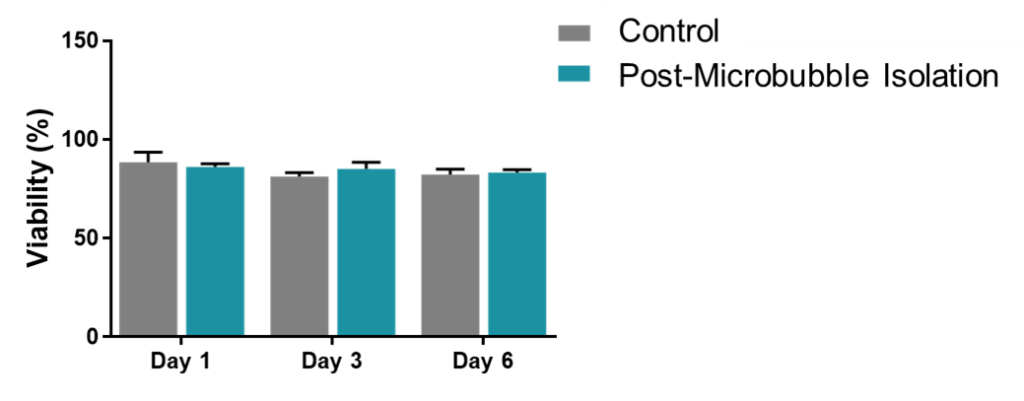
Using a quick and gentle cell separation method is a great way to ensure maximum viability. While magnetic activated cell sorting (MACS) and buoyancy-activated cell sorting (BACS) are among the quickest cell separation methods available, BACS represents a much gentler approach to cell isolation. Since MACS relies on much smaller particles than BACS, there is the potential that these particles could be taken up by target cells. Additionally, MACS relies on a strong magnetic field to pull the target cells away from the rest of the sample. The potential uptake of the magnetic particles coupled with the harsh forces associated with the strong magnetic field may lead to physiological changes that hide or distort the phenotype you are investigating.
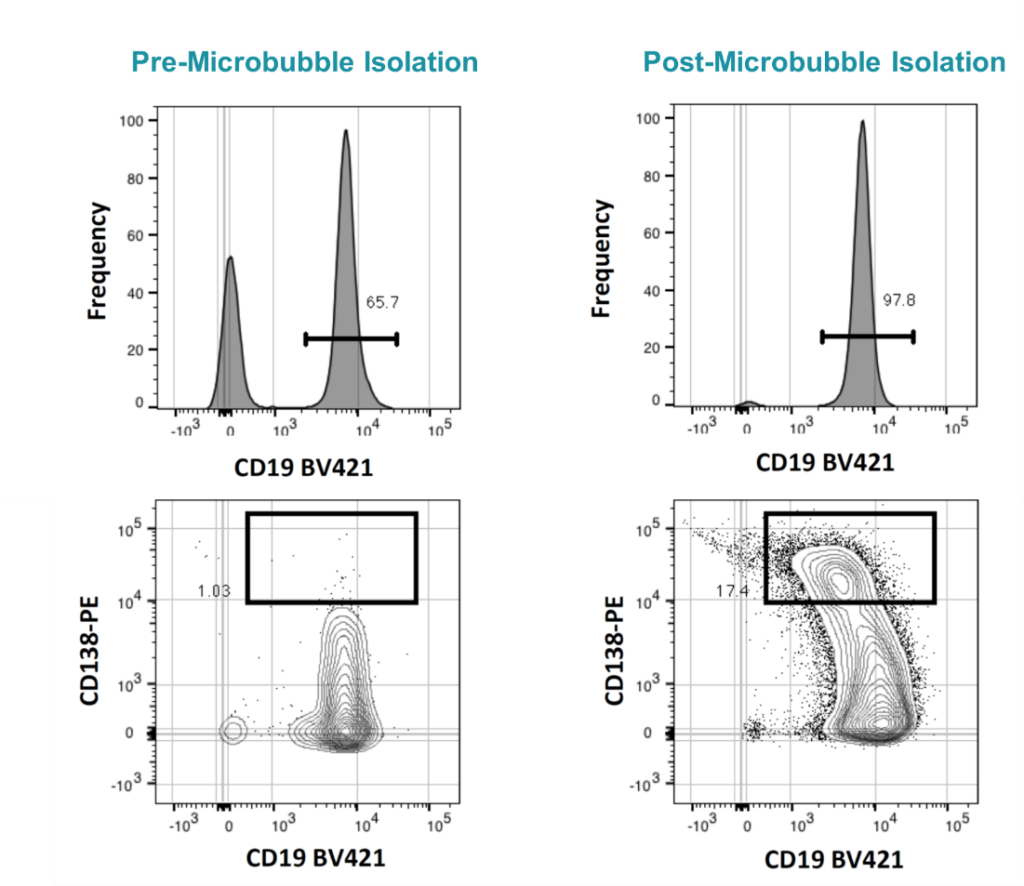
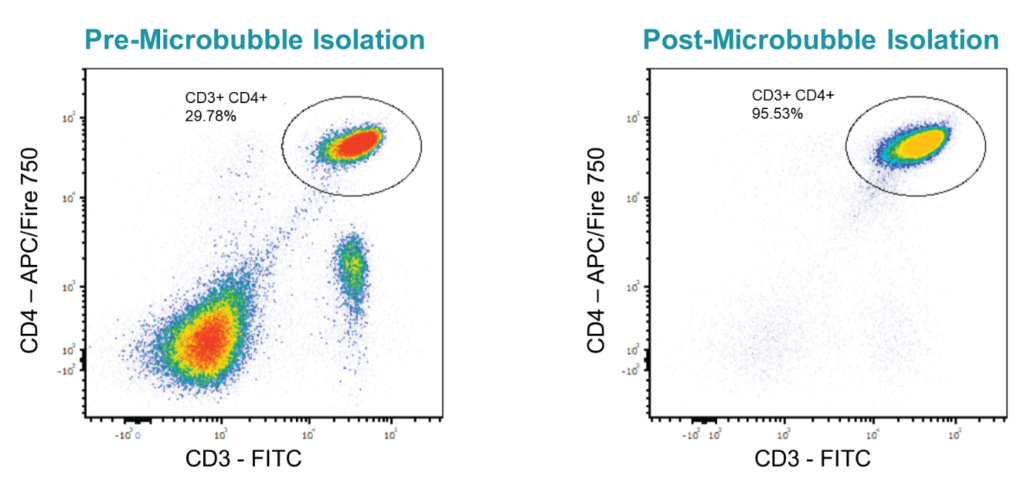
CD19+ B cells removed from mouse spleens were analyzed before (left) and after microbubble isolation (right). Cultured isolates were exposed to CpG and IL5 for three days and then plasma cell induction (CD138 upregulation) was assessed (bottom). Data courtesy of Derek Jones (University of Pennsylvania).
What Is the Importance of Consistency and Reproducibility in Cell-Based Research?
Reproducibility refers to the ability of researchers to obtain similar results from replicates of an experiment. If you or others have difficulty reproducing your results, then it will be a real challenge to reach statistical significance. Thus, reproducibility is a key to your ability to draw meaningful conclusion from your data.
To reduce variability, it is good practice to start each experiment with a high-quality sample, limit the length of the protocol to reduce the time that the sample is out, limit the number of steps in the protocol to increase efficiency and reduce the chance of making mistakes, and decrease the requirement for manual manipulation of your sample.
Why Consider the Speed of Cell Separation Protocols?
The speed of the separation protocol can influence the quantity and quality of the cells you are able to isolate, which in turn may affect reproducibility. Additionally, the longer it takes to complete the cell isolation protocol, the fewer samples you can process and less time you have for other lab tasks.
To maximize efficiency and throughput, a rapid and easy protocol is recommended. Buoyancy Activated Cell Separation (BACS) represents one of the quickest and easiest cell separation methods.

What Is the Value of a Simplified Cell Separation Method?
A self-standing cell separation kit that relies on only a few reagents and offers a straightforward protocol makes any isolation effortless.
Akadeum’s GMP-compatible microbubble technology represents a game changing method in cell isolation. We offer a simple and easy-to-follow protocol that allows for the entire cell isolation procedure to occur in a single tube. Our kits eliminate the need for additional equipment or advanced training and the potential for target cell loss associated with transferring samples from tube to tube.
Is There a Low-Cost Technique for Isolating Cells?
The scarcity of research funding makes cost a major factor in lab decisions. Since Akadeum’s microbubbles work with common lab materials and don’t require you to purchase expensive magnets or columns, they represent an excellent low-cost cell separation method.