An In-Depth View of Autologous Cell Therapy
The development of adoptive cell therapies like CAR T-cell therapy and tumor-infiltrating lymphocyte (TIL) therapies revolutionized the cancer treatment sector of the biopharmaceutical industry. These therapies steer the human immune system to fight and eradicate cancers.
What Is Autologous Cell Therapy?
The groundbreaking development of autologous therapy brought new interest to cell therapy initiatives. Autologous cell therapy utilizes healthy cells from a patient’s peripheral blood to treat disease or replace damaged bone marrow after chemotherapy, radiation, or other deteriorative cancer treatments.
By extracting the blood components directly from the patient themselves, autologous cell therapy diminishes the risk of incompatibility between donors and patients. Patients with sufficient cell supply for apheresis qualify for this treatment.
In many cases where a patient is facing a severe diagnosis, tumor-targeting treatments like chemotherapy are unavoidable and can damage blood-forming cells in the bone marrow.
Mobilization factors can boost stem cell counts within the patient’s peripheral blood. Once the periphery is rich with these cells, leukapheresis gathers the patient’s leukocytes. Leukapheresis is a medical procedure that harvests white blood cells from whole blood and returns the white-blood-cell-depleted blood to the patient.
Leukocytes contain a high concentration of T cells, and this cell population is isolated from the sample for modification, activation, and expansion. Explore Akadeum’s T cell isolation kit to discover easy cell isolation methods utilizing innovative microbubbles for cell therapy technology.
Once an adequate number of high-quality T cells are isolated, they are activated to trigger ex vivo expansion. Expansion is triggered through the co-stimulation of CD3 and CD28 at the MHC I molecule after a specific antigen binds to the T cell, a phenomenon called T cell activation. Cytokines added to ex vivo expansion protocol encourage activation and rapid expansion. Discover how easy ex vivo expansion can be with Akadeums T cell expansion protocol. The expanding T cells are also genetically modified to express CARs, which directly target cancerous cells. The CAR T cells are tested for quality and cryopreserved for return to the treatment facility. Once they arrive, the T cells are thawed and returned to the patient via infusion.
CAR proteins are composed of an extracellular domain—often designed based on a monoclonal antibody specific to tumor cells—and an intracellular domain similar to the signaling domains of T cell activating receptors. This design allows the CAR T cell to kill tumors efficiently, live longer in vivo, and function safely and effectively.
Because these cells originated within the patient, there is minimal risk of graft-versus-host disease, which can be prevalent in therapies that utilize healthy donors. However, autologous CAR T-cell therapy is highly personalized, and one batch of CAR T cells serves only a single patient, presenting scaling issues.
T Cell Expansion of Cells Extracted Via Apheresis
Autologous cell therapy harvests a patient’s T cells for clinical application via apheresis. This approach allows for a specified and specific treatment with a reduced risk of immune rejection.
Because the patients undergoing this apheresis are typically severely ill, the T cells are mature, differentiated, and may have stunted growth rates. Highly matured cells can present a significant problem to ex vivo expansion protocols. Damaged or weakened mature T cells offer a lower-quality cell population than healthy cells and can slow CAR T-cell expansion. Patient cells are also often functionally weaker cytotoxically, meaning they have a feeble tumor-killing ability.
Steps of an Autologous Car-T Workflow
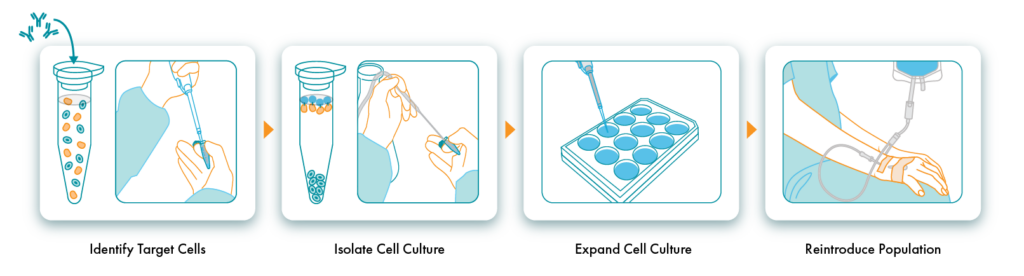
Identify Target Cells
In autologous cell therapies, cells are harvested directly from the patient needing therapy and targeted via leukapheresis. In addition to T cells, different blood components are being explored for cell therapy, including B cells, NK (natural killer) cells, and dendritic cells. Autologous CAR T-cell therapy targets and selects healthy T cells within the patient’s peripheral blood.
Isolate and Expand Cell Culture
The leukapheresis process captures many types of white blood cells, many of which are T cells. These T cells can be isolated using cell separation techniques. Most techniques implement positive T-cell selection, tagging the T cells and removing them from the solution using antibodies.
Akadeum offers an innovative cell separation approach using our specialized microbubble technology, antibodies, and negative selection. The final T cell population is robust and untouched by negatively selecting unwanted cells.
After successfully isolating a pure sample of T cells, they are activated and expanded using activation-inducing reagents. The expansion step is crucial to successful autologous cell therapy and can be challenging to achieve with weakened patient T cells. Inadequately expanded T cells will not provide effective adoptive T-cell therapy.
Reintroduce Population
Once T cells have been isolated, activated, and expanded, they are reintroduced into the patient’s body via intravenous infusion.
Challenges of Autologous Cell Transplantation
Autologous cell therapies, though revolutionary, present multiple disadvantages to commercial availability. These include scaling difficulties, expansion reliability, and cost.
Autologous cell therapy uses patient cells, which provide a hyper-specific and personalized treatment design. This design could be more conducive to a large-scale treatment initiative, as each patient must undergo apheresis to obtain an adequate quantity of T cells.
Autologous therapy can take up to a month, and delays in this process can be devastating for terminally ill patients. And the heightened cost of autologous cell therapy is an obstacle to its accessibility. One treatment can range between $370,000 to $475,000, a steep price for a treatment with such potential for failure.
Autologous cell therapy can also lead to serious side effects, such as cytokine release syndrome (CRS), where the reintroduced cells cause an uncontrollable release of cytokines, driving an unstoppable immune response. If not correctly and quickly addressed, this can have catastrophic effects on a patient, like significant organ failure and death.
Autologous vs. Allogeneic Cell Therapy
Patient successes with autologous cell therapy ignited interest in other cell therapies, and soon the development of allogeneic cell therapies was underway. Allogeneic cell therapy attempts to resolve many of autologous cell therapy’s scaling and cost issues by using healthy donor cells as a T-cell source for activation and expansion, as opposed to patient cells.
The hope of Allogeneic cell therapy is that it will be more accessible to patients than autologous therapy because of its cost and easier logistics.
Using Akadeum’s Cell Expansion Technology to Increase Sample Viability
With any form of cell therapy, the end-product quality and quantity depend on the initial population of T cells activated and expanded ex vivo. Many studies have shown that higher-quality T-cell inputs are necessary for robust expansion leading to long-lived and effective cell therapy treatment. Using inadequate starting material can considerably impact downstream manufacturing.
As part of a broader focus on cell therapy workflow optimization, Akadeum provides straightforward and reliable T cell isolation, so the cell therapy starting material is optimal every time. After harvesting patient leukocytes, ensure successful T cell activation and expansion by isolating T cells using Akadeum’s Human T cell Isolation Kit. Using a negative selection method, T cells can be isolated free of harm from separation beads and off-target effects of antibodies, leaving the solution rich with untouched T cells and ready for activation and expansion.



