Immune Control and Evasion During Acute and Chronic HBV Infection
Updated on Oct 16, 2024 Share
Investigating the role of T cell exhaustion and T cell memory in hepatitis B virus infections
Dr. Hannah Drescher, a Postdoctoral Research Fellow in Professor Georg Lauer’s lab at Mass Gen Hospital and Harvard Medical School, used Akadeum’s Human CD4+ T Cell Isolation Kit with BACS™ Microbubbles to isolate cells to study the role of T cells during acute and chronic HBV infection. Specifically, Hannah is interested in studying the correlates of protection, as well as the mechanisms employed by viruses to escape host immune surveillance during chronic viral infection. The lab’s goal is to develop immunotherapeutic interventions that restore T cells from T cell exhaustion, a state of dysfunction that causes poor effector T cell response during chronic viral infection.
The research group has established innovative approaches that allow them to overcome research challenges in studying immunological mechanisms of action during viral infections. Already, they have successfully generated genotypic and phenotypic profile candidates using microarray, scRNA-seq and ATAC-seq data from virus-specific CD4+ and CD8+ T cells in blood and liver to elucidate the specific molecular pathways associated with viral control and immune failure.
Antigen-specific (HBV-specific) CD4+ T cells are of exceptionally low abundance in persistent HBV infections, representing roughly 0.01% of the original CD4+ population. In order to progress with her groundbreaking work, Hannah required a cell isolation method that reliably delivers high yields of high-quality cells without sacrificing purity.
The BACS™ Microbubble approach delivered on their needs by producing more cells at a higher purity than a magnetic counterpart. All of this was accomplished while being exceptionally gentle and eliminating the need to expose cells to harsh chemicals, magnetic, or shear forces.
Objective
Obtain a high yield of pure CD4+ T cells from human PBMCs of chronically infected individuals.
Kit Used: Human CD4+ T Cell Isolation Kit from Akadeum Life Sciences
The Human CD4+ T Cell Isolation Kit contains a cocktail of biotinylated antibodies and streptavidin-conjugated BACS™ Microbubbles to target and remove unwanted cells.
Procedure
Thirty million frozen patient derived PBMCs were thawed. CD4 negative cells were labelled with an optimized cocktail of antibodies targeting CD8, CD11b, CD14, CD16, CD19, CD20, CD36, CD56, CD123, CD161, CD235ab, CD314, and γδTCR, and subsequently mixed with BACS™ Microbubbles. The recommended volume of reagents was added based on total cell number. Samples were centrifuged to separate CD4+ T cells from microbubble-laden RBCs and CD4 negative mononuclear cells which collect at the surface and were subsequently removed by vacuum-aspiration. The remaining CD4+ T cells were collected and used in downstream workflows. Tetramer staining was used to identify antigen-specific T cells.
Outcome
In a head-to-head comparison with a magnetic bead-based protocol, the Akadeum protocol delivered 95.4% purity as compared to 86.7%. The enriched CD4+ T cell sample subsequently enabled Hannah to effectively isolate the low abundant HBV-specific CD4+ T cells.
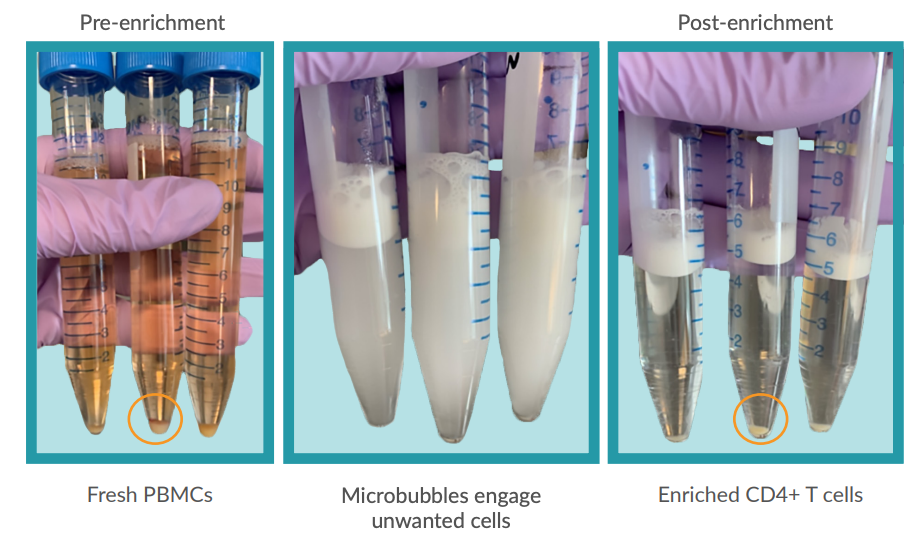
Figure 1: BACS™ Microbubbles Enable Fast and Easy Sample Enrichment (left) PBMC starting sample from HBVinfected patients. (center) Samples containing biotinylated-antibody cocktail and BACS™ Microbubbles. (right) Samples after centrifugation showing BACS™ Microbubbles collected at the surface and cell pellet containing untouched CD4+ T cells.
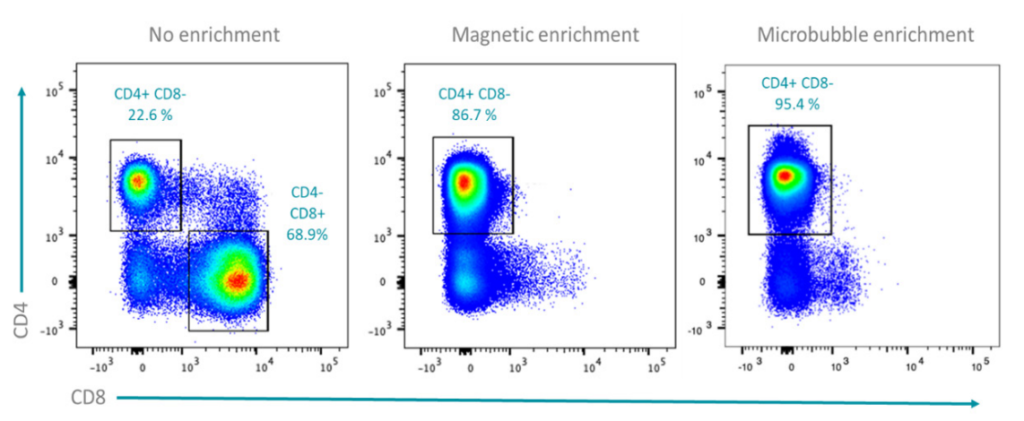
Figure 2: Comparative Study
PBMCs (left), CD4+ T cells enriched with magnetic bead-based isolation (center), and CD4+ T cells enriched with BACS™ Microbubbles (right). In PBMCs of viral-infected patients, CD4+ T cells accounted for 22.6% of the sample. Standard magnetic bead-based enrichment resulted in a final purity of 86.7%, while BACS™ Microbubble enrichment resulted in 95.4% purity.
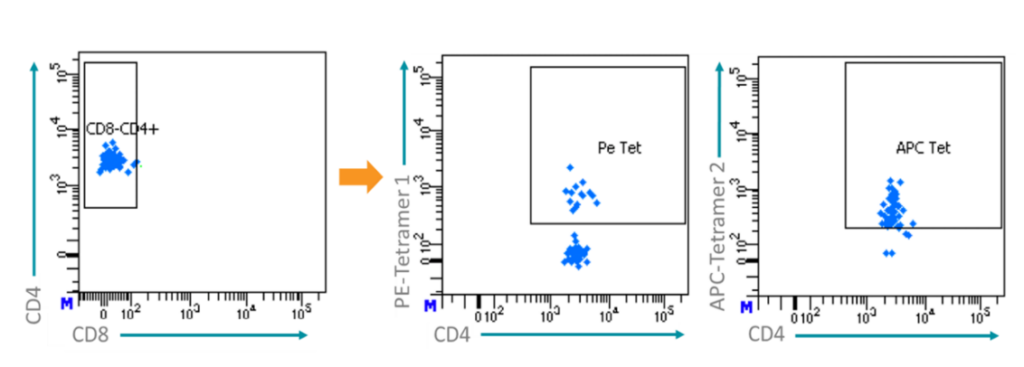
To identify HBV-specific CD4+ T cells tetramer staining was used. Patient-derived T cells that recognize HBV-specific epitopes were labeled, detected, and collected. Two separate HBV-specific epitopes attached to fluorophores, one labeled with PE (center) and the other with APC (right), were used.


